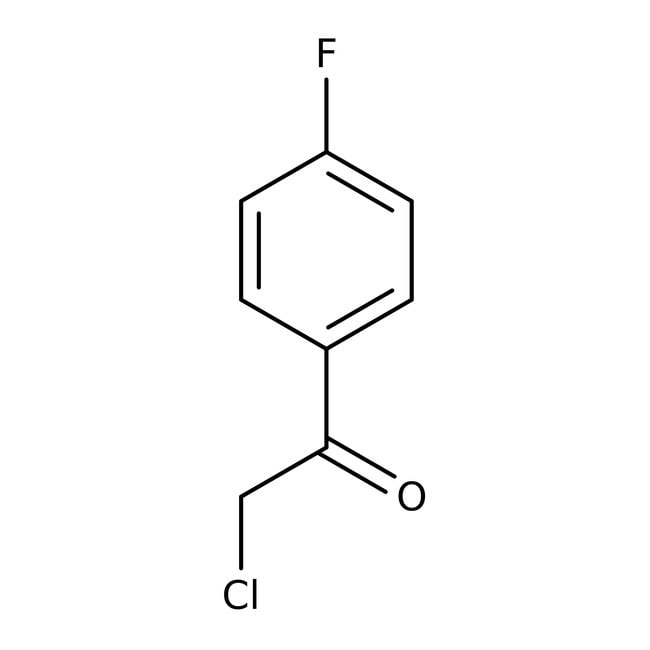
EP0002151A1 - Process for the synthesis of esters of parachlorobenzoyl-phenoxy-isobutyric acid - Google Patents

Condensation de Claisen Réaction de condensation Réaction chimique Acétoacétate d'éthyle Ester, autres, angle, texte png | PNGEgg
Benzoic Acid Molecule Isolated on White Stock Illustration - Illustration of structure, additive: 43832779

EP0074309A1 - Process to increase the tolerance of opacifying products, and opacifying products so obtained - Google Patents

Quelques produits de transformation de l'acide 4, 5‐dinitro‐2‐chloro‐ benzoïque - Goldstein - 1938 - Helvetica Chimica Acta - Wiley Online Library

EP0074309A1 - Process to increase the tolerance of opacifying products, and opacifying products so obtained - Google Patents
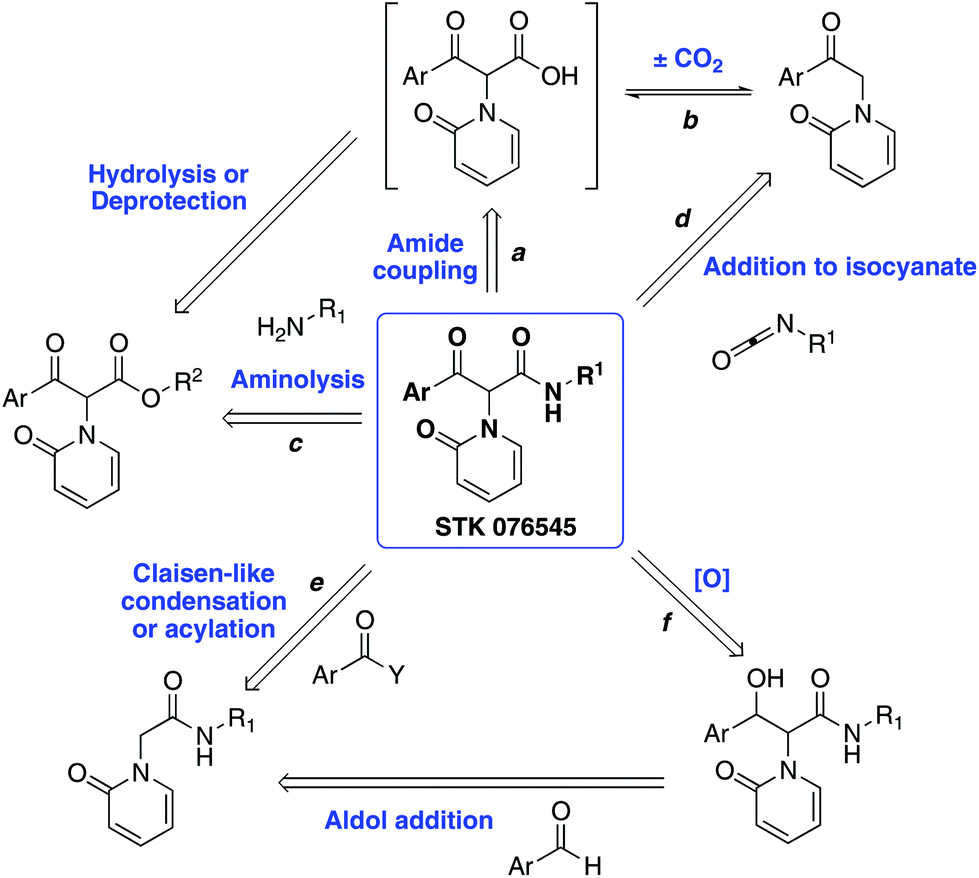
Route exploration and synthesis of the reported pyridone-based PDI inhibitor STK076545 - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB01205J

EP0002151A1 - Process for the synthesis of esters of parachlorobenzoyl-phenoxy-isobutyric acid - Google Patents

How would you prepare the given compound from toluene? A diazonium replacement reaction is needed in some instances. | Homework.Study.com

Starting with cyclohexanone, show how to prepare these compounds. In addition to the given starting material, use any additional organic or inorganic reagents that are required (a) 1-Phenylcyclohexene (b) Cyclohexene oxide (c)






![sublimation de l'acide benzoique - [BTS métiers de la chimie] sublimation de l'acide benzoique - [BTS métiers de la chimie]](https://www2.ac-lyon.fr/etab/lycees/lyc-69/martiniere-diderot/BTSchimie/local/cache-gd2/e16261c75390c3fa5c50ea15a2de0183.jpg)