Would a solution with a [H+] concentration of 1 X 10-11 have a pH value of ___ and be considered a(n)? - Quora
The pH of a solution describes its acidity and is the negative logarithm ( log) of its hydrogen ion concentration. The term pH is used because the hydrogen ion concentration in solutions of weak acids and in many other fluids is frequently much less than 1 ...
![pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom](https://i.pinimg.com/736x/99/80/05/998005d7b3fbb74f7a91222f3209e7c5--physical-chemistry-ap-chemistry.jpg)
pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom
![SOLVED: Acidic or basic solution, pH The pH of a solution is the negative logarithm of the hydrogen-jon concentration: pH = #log[h"] log stands for logarithm to the base 10 log 100 = SOLVED: Acidic or basic solution, pH The pH of a solution is the negative logarithm of the hydrogen-jon concentration: pH = #log[h"] log stands for logarithm to the base 10 log 100 =](https://cdn.numerade.com/ask_images/54471c304890403dbf6cbad11744b807.jpg)

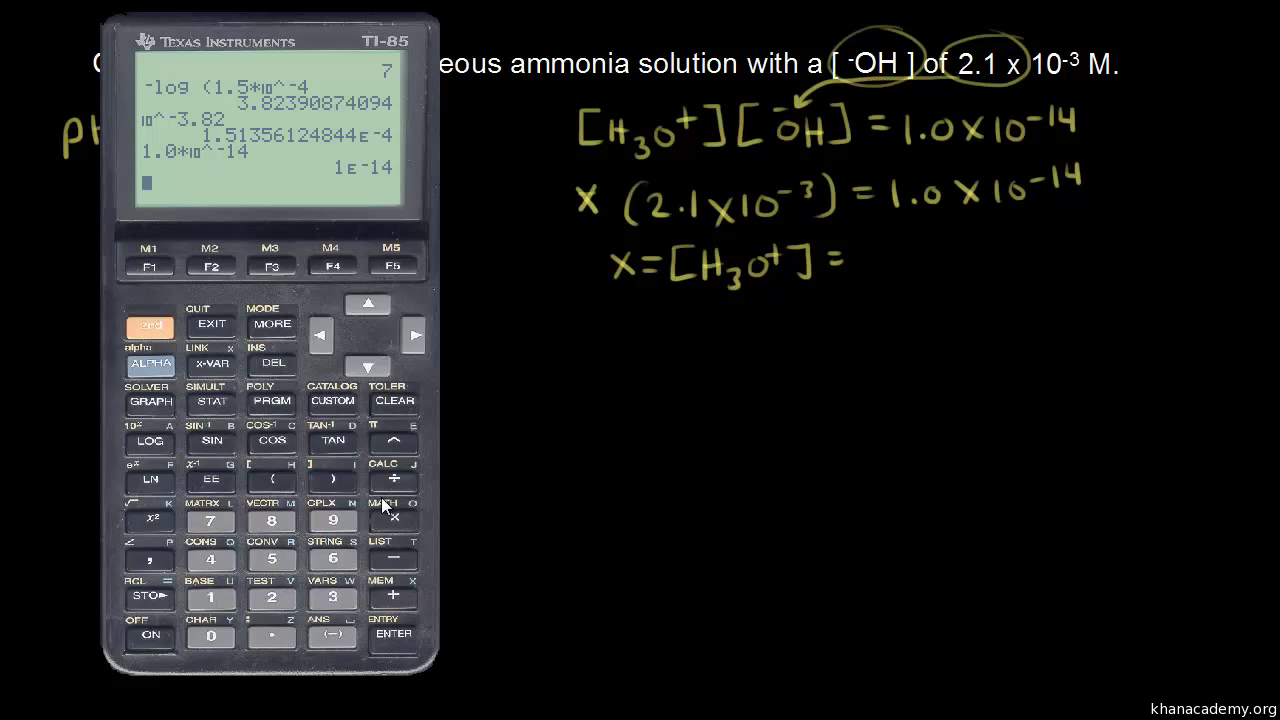



![Given pH & pOH, Solve for [H+] & [OH-] Practice Problems - YouTube Given pH & pOH, Solve for [H+] & [OH-] Practice Problems - YouTube](https://i.ytimg.com/vi/Pm99tsEOyGQ/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AHCB4AC0AWKAgwIABABGGAgYChgMA8=&rs=AOn4CLBiQ7kUcjmLXRL3xjFxt985w-xdoQ)

![Слайд 24 pH of Common Substances Слайд 25 Calculating the pH pH = - log [H+] (Remember that the [ Слайд 24 pH of Common Substances Слайд 25 Calculating the pH pH = - log [H+] (Remember that the [](https://www.sliderbase.com/images/referats/1143b/(25).PNG)

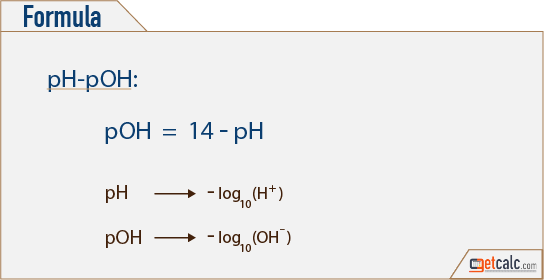

![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)
:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)

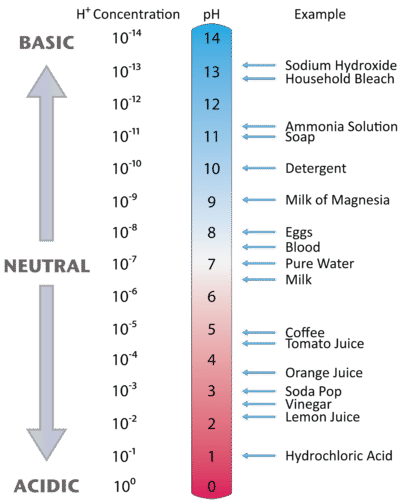
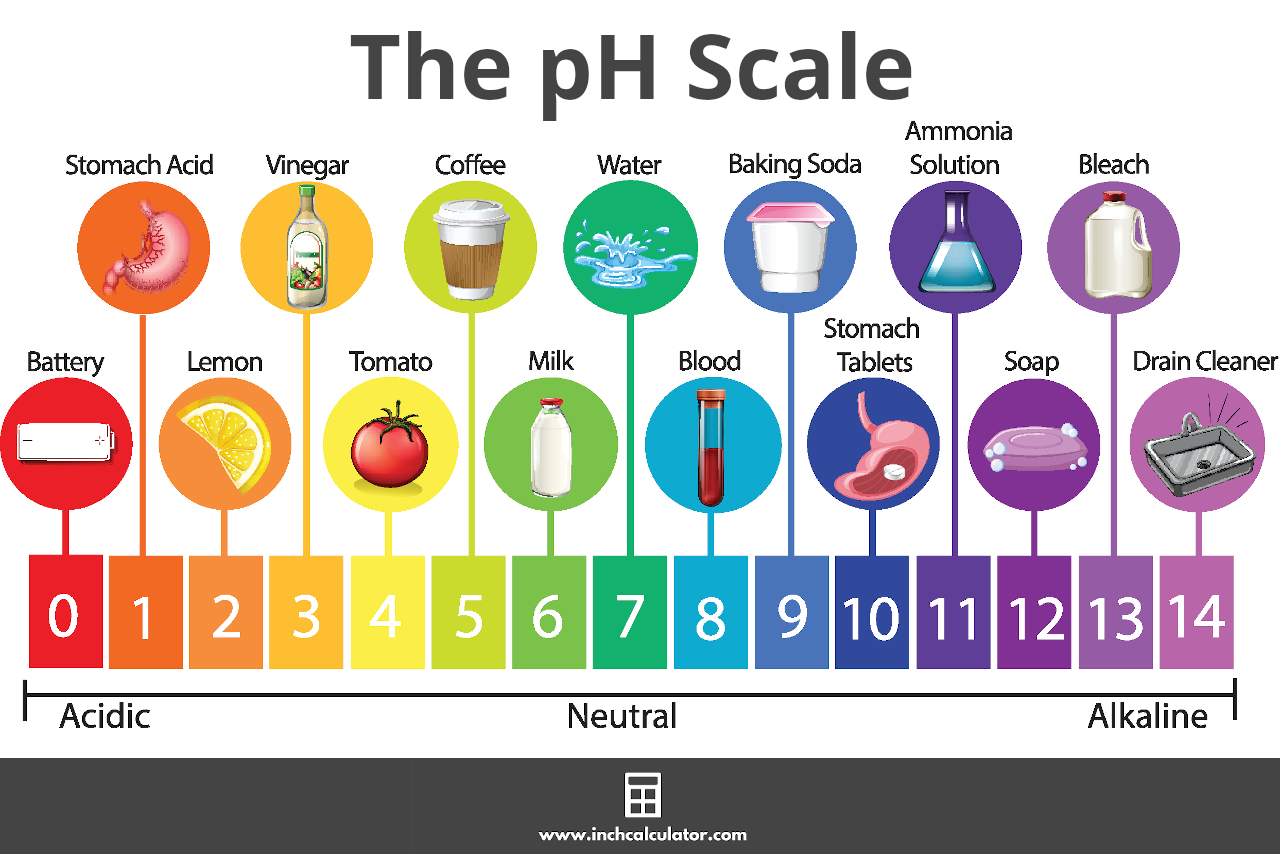

![pH = -log[H+] pH = -log[H+]](https://s3.studylib.net/store/data/008427242_1-6a856521ca6b3bcc442fa00c77f4fe6e.png)


![Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC" Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC"](http://itsactuallyquitebasic.weebly.com/uploads/2/7/8/0/27808159/4950515.png?357)